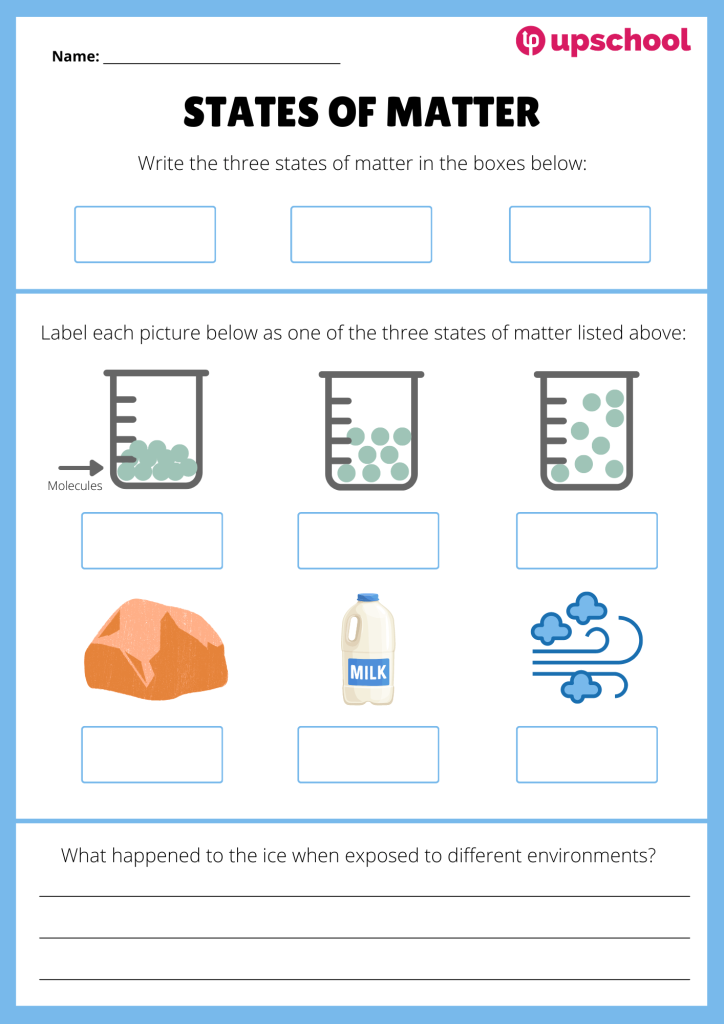
Liquid, solid, gas! These three states of matter all behave very differently, and today we will explore why.
The difference in the arrangement of particles in each type of matter explains why ice, water, and water vapour all look different, despite being made up of the same molecules. Solids, such as ice, have particles that are tightly packed together, while liquids and gases have particles that are farther and farther apart, allowing them to move more freely.
How do we transform a substance from one state to another? We apply or remove heat!
Download the resource below to help your students learn to identify each state of matter. For extra practice, try doing an in-class demo showing your students how ice can be heated to become liquid water or water vapour!
Resource Preview: